Background and Drug Introduction
Background
When it comes to hypertension treatment, the norm for patients has long been "daily medication and lifelong management". However, poor compliance and unstable blood pressure control have always been major clinical pain points. Statistics show that China has a huge population of hypertension patients. The inconvenience caused by long-term medication often leads some patients to stop or reduce medication without medical advice, thereby increasing the risk of cardiovascular and cerebrovascular complications. The advent of MWX401 Injection is expected to completely change this situation. Relying on the unique advantages of siRNA technology and combining the targeted delivery capability of nanobodies, it pioneers a new "precise, long-acting, convenient and hassle-free" approach for hypertension treatment.
MWX401 Injection
MWX401 Injection is a Class 1 innovative chemical drug with global intellectual property rights. Its clinical trial application was submitted to the Center for Drug Evaluation (CDE) of the NMPA in December 2025 and approved after a rigorous review in just three months. This not only demonstrates the high-efficiency progress of innovative drug R&D in China but also reflects the profound domestic accumulation in the fusion technology of siRNA and nanobodies. The approval of this novel drug not only enriches the innovative approaches for hypertension treatment but also marks a key step in the application of nanobodies in the delivery of nucleic acid drugs, providing a new direction for the R&D of therapeutic drugs for chronic diseases.
Core Technologies
The "Molecular Scissors" Effect of siRNA:
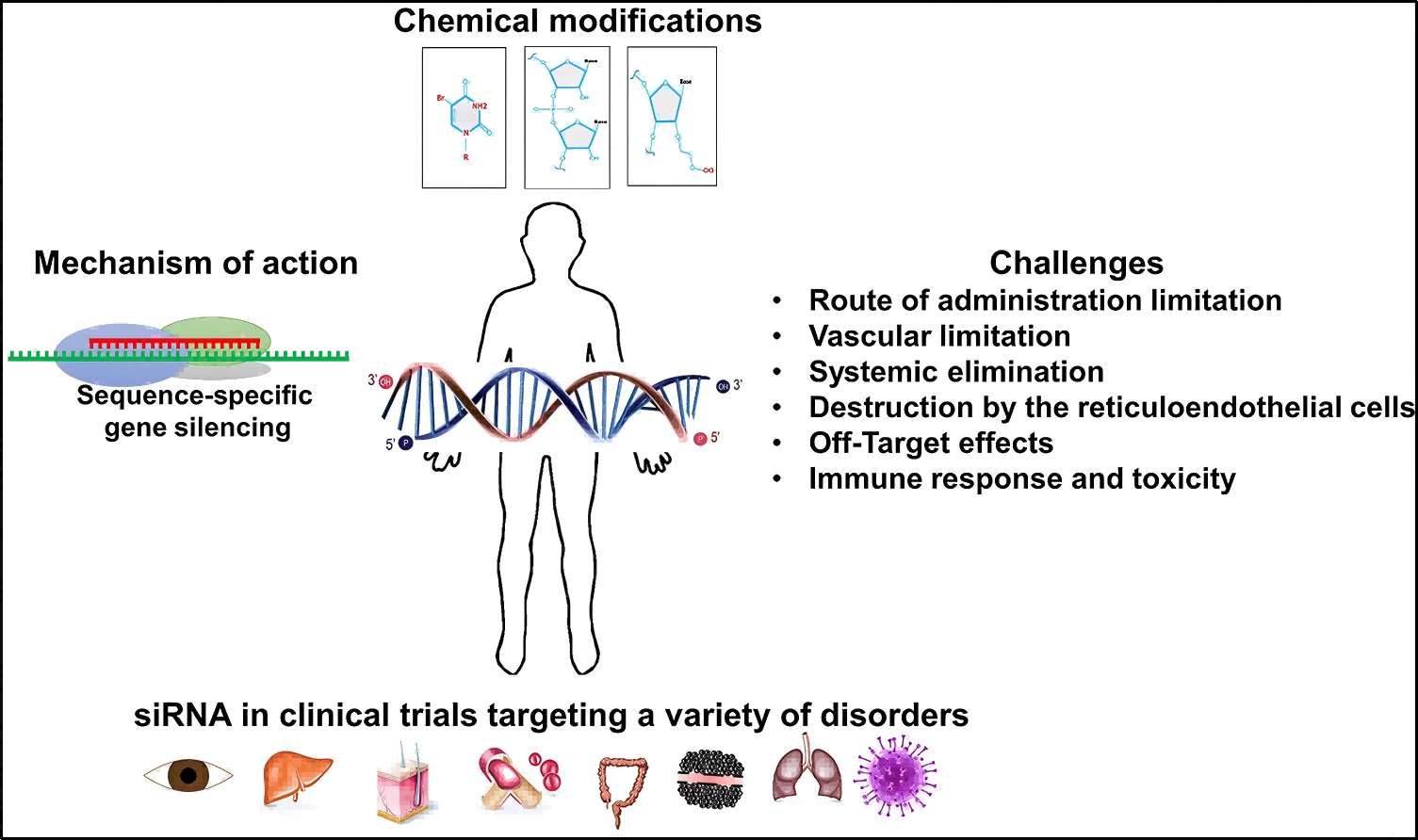
As a mainstream drug form of the next generation, siRNA drugs stand out for their mechanism of "precise inhibition". Acting like a pair of molecular scissors, siRNA specifically recognizes and cleaves the mRNA of target genes through the RNA interference mechanism, blocking the production of angiotensinogen (AGT) — a pathogenic protein — at the source. As the upstream initiating molecule of the renin-angiotensin-aldosterone system (RAAS), the inhibition of AGT synthesis can completely block the activation of the RAAS pathway (one of the core mechanisms underlying hypertension), thus achieving a curative rather than symptomatic therapeutic effect. Compared with traditional small molecule drugs and antibody drugs, siRNA drugs have distinct advantages of high efficiency, long-acting efficacy and high specificity.
Figure 1. Mechanism of action of siRNA therapy[1]
The "Precision Delivery" Effect of Nanobodies:
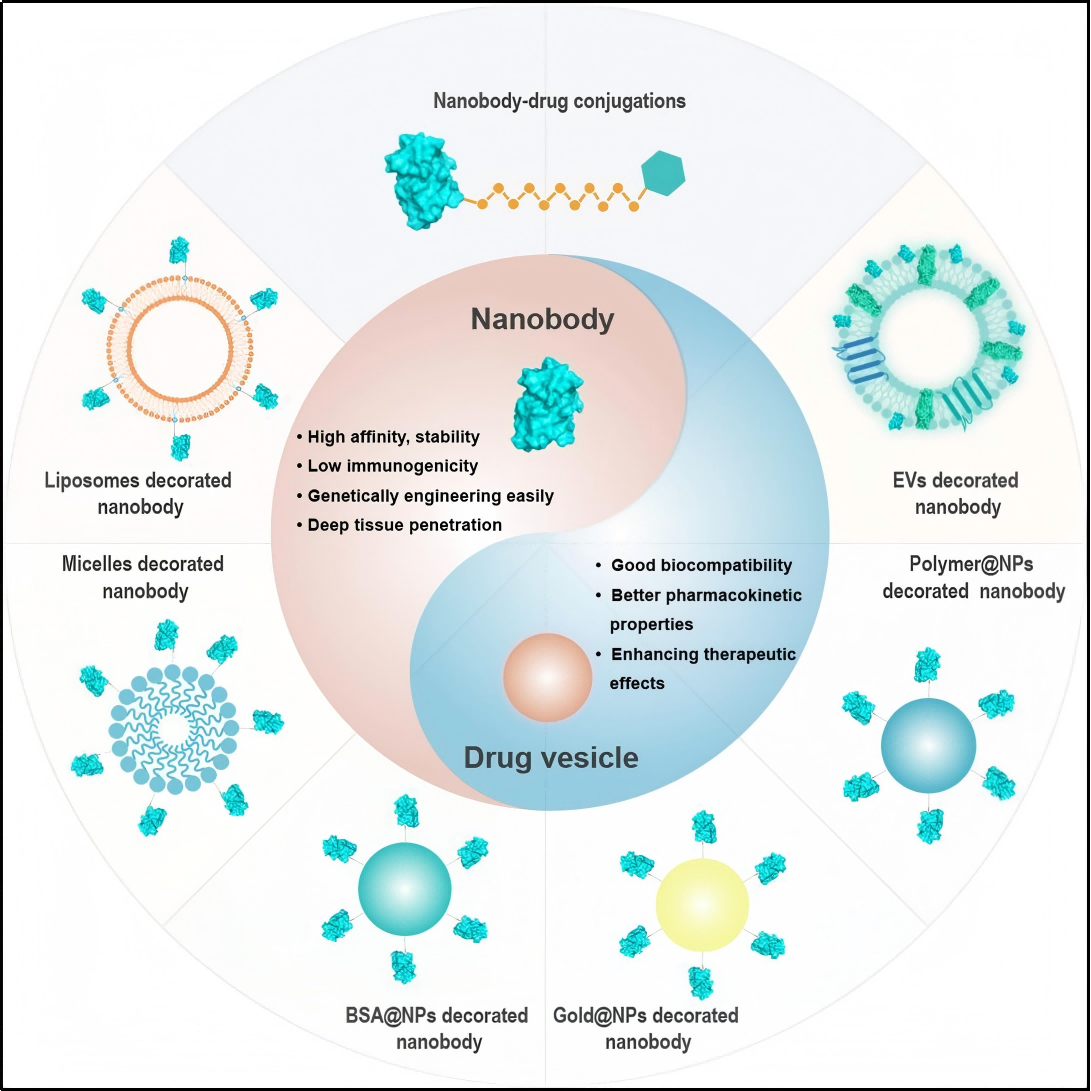
Nanobodies are a new type of molecular format antibodies with small size, high stability, strong specificity and low immunogenicity, which have been widely applied in targeted drug delivery for cancer therapy. Their core function is precision delivery: they can accurately recognize and bind to hepatocytes, delivering the conjugated siRNA drugs precisely to the liver — the major site of AGT protein synthesis. This reduces drug distribution in extrahepatic tissues, lowers the risk of off-target effects and systemic side effects, while improving drug delivery efficiency.
Figure 2. Nanobody-based drug delivery systems[2]
Preclinical Date and Safety
Impressive Preclinical Data
Non-clinical study results showed that in humanized hypertensive mice, MWX401 significantly reduced the serum levels of the target protein and its mRNA, while effectively lowering systolic and diastolic blood pressure. In spontaneously hypertensive rhesus monkeys, a single subcutaneous administration of MWX401 Injection at different doses not only markedly decreased the animal's AGT levels but also significantly reduced systolic and diastolic blood pressure, presenting a favorable dose-effect relationship, and its antihypertensive effect was superior to that of valsartan, the positive control drug. Notably, the antihypertensive effect was sustained until the end of the trial, with the pharmacodynamic effect lasting for at least 12 weeks in animals. This means patients only need to receive medication four times a year, reducing the dosing frequency by more than 99% compared with daily oral medication, which will greatly improve patients' treatment compliance.
Safety
MWX401 Injection also demonstrated excellent safety profiles, which is also attributable to the incorporation of nanobodies. Safety evaluation trials showed that the drug had no unexpected safety signals. The precise delivery characteristic of nanobodies ensures that siRNA is mainly distributed in the liver with limited presence in extrahepatic tissues, which greatly reduces the risk of off-target effects and systemic side effects, providing a reliable safety guarantee for subsequent clinical trials and future clinical applications.
Industrial Significance
Behind the IND approval of MWX401 Injection lies not only an important extension of siRNA technology from the field of rare diseases to chronic diseases but also a pivotal breakthrough in the integrated application of nanobodies and nucleic acid drugs. Previously, siRNA drugs have been successfully validated in the treatment of hypercholesterolemia, hypertriglyceridemia and other conditions. The integration of nanobodies has further expanded the application boundary of siRNA drugs. This time, MWX401 extends this fusion technology pathway to hypertension — a chronic disease with a larger patient population and more prominent compliance challenges, marking a solid step in China's exploration of the synergistic application of nucleic acid drugs and nanobodies in chronic disease management.As China's first AGT siRNA antihypertensive drug integrated with nanobodies, MWX401 Injection has a significant first-mover advantage. The advancement of its subsequent clinical trials will not only provide a higher-quality and more convenient treatment option for hypertension patients but also further drive the fusion and innovation of nanobody and siRNA technologies, enhancing the international competitiveness of domestic innovative drugs.
Referuence:
- Liu L, Tu B, Sun Y, Liao L, Lu X, Liu E, Huang Y. Nanobody-based drug delivery systems for cancer therapy. J Control Release. 2025 May 10;381:113562. doi: 10.1016/j.jconrel.2025.02.058. Epub 2025 Feb 22.
- Alshaer W, Zureigat H, Al Karaki A, Al-Kadash A, Gharaibeh L, Hatmal MM, Aljabali AAA, Awidi A. siRNA: Mechanism of action, challenges, and therapeutic approaches. Eur J Pharmacol. 2021 Aug 15;905:174178. doi: 10.1016/j.ejphar.2021.174178. Epub 2021 May 24. Erratum in: Eur J Pharmacol. 2022 Feb 5;916:174741.
- Zhang Y, et al. Small but mighty: the versatility of nanobodies in modern medicine[J]. RSC Advances, 2025, 15(2): 1024-1042.
- Li Y, et al. Emerging insights and future prospects for therapeutic application of siRNA targeting angiotensinogen in hypertension[J]. Pharmacological Research, 2023, 195: 106881.

Wuhan Nanobody Life Science & Technology Co., Ltd. is a nanobody industry platform established under the lead of the Wuhan Industrial Innovation & Research Institute. Headquartered in the main building of the Institute within the East Lake High-Tech Development Zone in Wuhan, the company operates a proprietary laboratory spanning 1,400 sq.m at the Precision Medicine Industrial Base in Wuhan Bio-City. It has also established alpaca experimental and transfer bases in Zuoling, Wuhan, and Tuanfeng, Huanggang, compliant with laboratory animal standards. Currently housing over 600 alpacas, these bases can provide alpaca immunization services with "zero immunological background" assurance for research institutions and antibody drug R&D enterprises.
Nanobody Life focuses on the development, engineering, and application of nanobodies, committed to building an integrated industry-academia-research experimental public service platform. It currently possesses a full-chain technology platform encompassing antigen preparation (peptides, proteins, and RNA), antibody discovery and engineering, and biological function verification/screening. RNA antigens include those structurally and sequentially optimized for alpacas. Antibody discovery and engineering services employ multiple technological routes, including phage display and mammalian cell display. Through the complementary use of multiple platforms, it provides flexible antibody discovery and engineering services for pharmaceutical companies and research institutes, facilitating drug and reagent R&D.
- Contact Information:
Phone: 400-822-9180
Email: marketingdept@nanobodylife.com